1 / 5










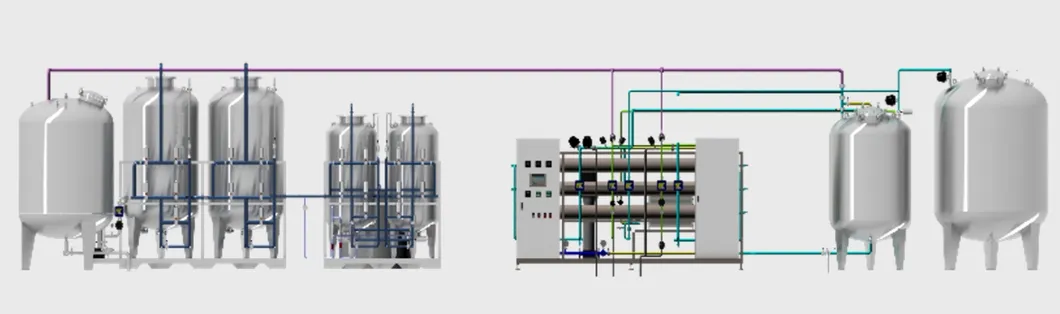
The 6000L/H purified water system is a cutting-edge solution meticulously crafted for the pharmaceutical, biotechnology, and healthcare sectors. It is engineered to rigorously adhere to both GMP and FDA standards, ensuring the reliable production of superior quality purified water essential for critical applications.
Seamlessly integrating advanced pretreatment, reverse osmosis, and optional EDI modules, this system provides efficient, energy-saving, and continuous operations.




| Item | Parameter |
|---|---|
| Capacity | 6000 L/H |
| Raw Water Source | Tap water / Groundwater |
| Material | SUS304 / SUS316L |
| RO Membrane | Imported brand |
| Control System | PLC + Touch Screen |
| Output Water Standard | USP / EP / ChP |