



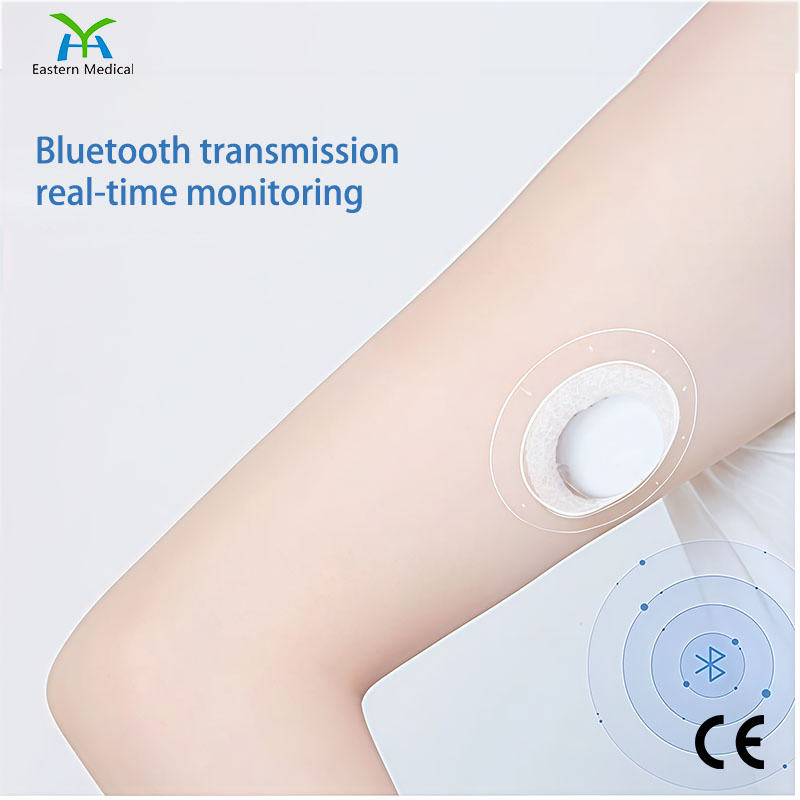
1 / 5











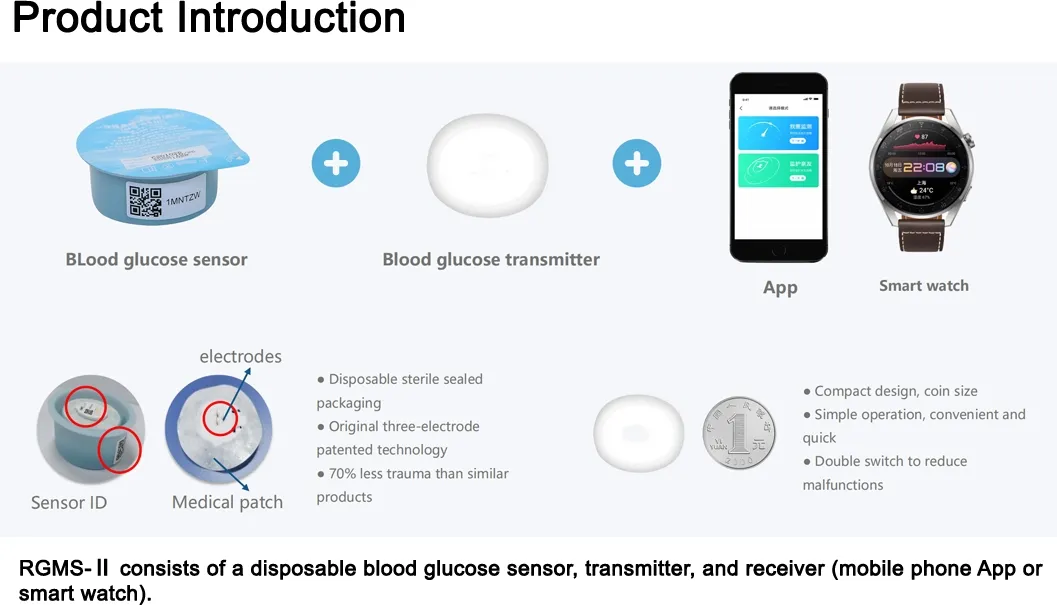

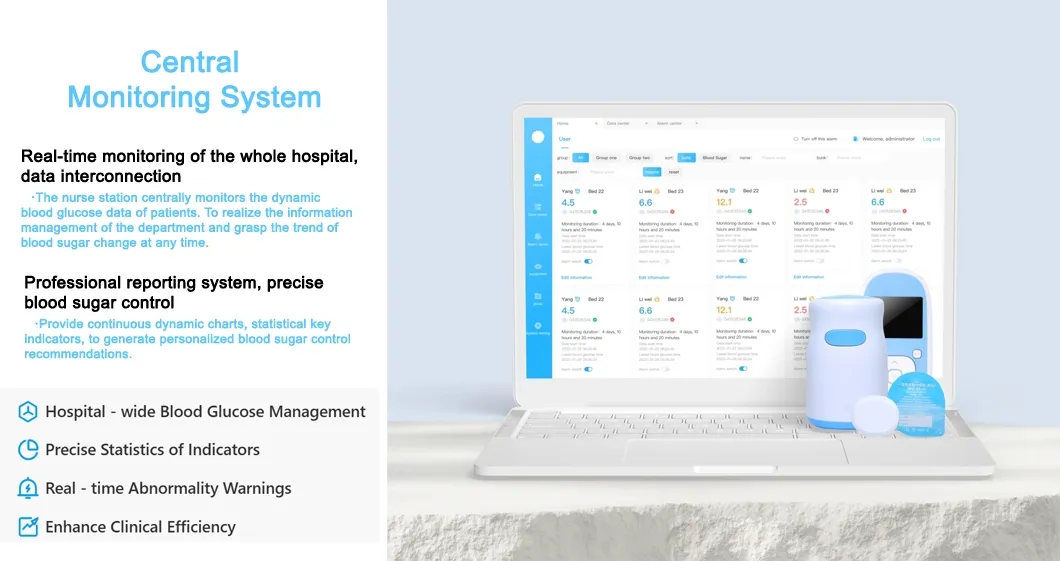
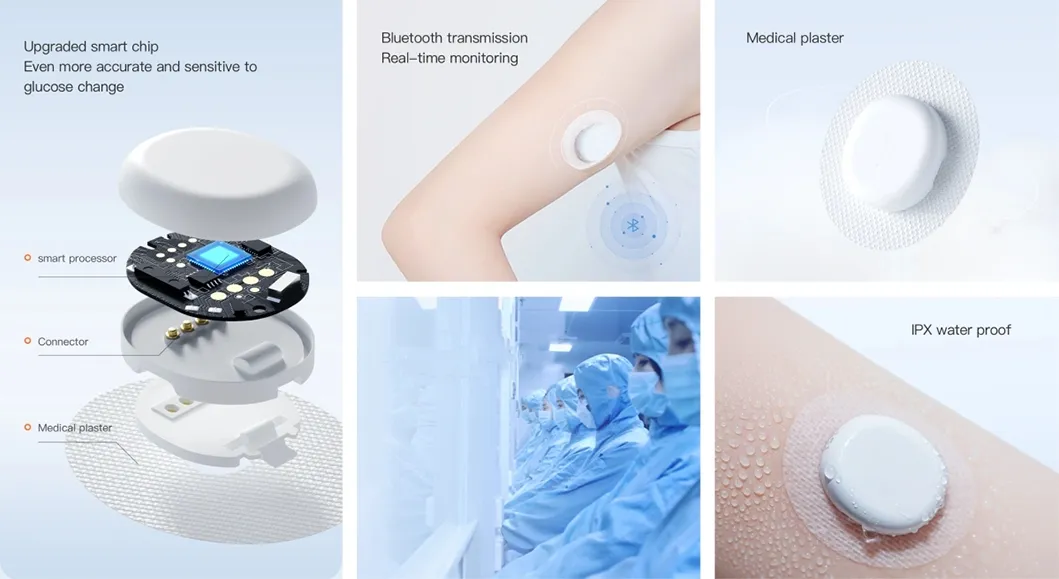




We are a dynamic and innovative medical equipment trading partner specializing in the global distribution of high-quality medical products. We are committed to sourcing, supplying, and supporting cutting-edge medical devices tailored to diverse clinical needs.
Our Core Focus:
Why Choose Us?
We prioritize understanding market needs to deliver tailored solutions. Through optimized supply chains, we offer cost-effective products without compromising quality. From pre-sale consultation to after-sales service, our team ensures seamless transactions and fosters long-term partnerships.